|
3/31/2024 0 Comments Periodic table molar masses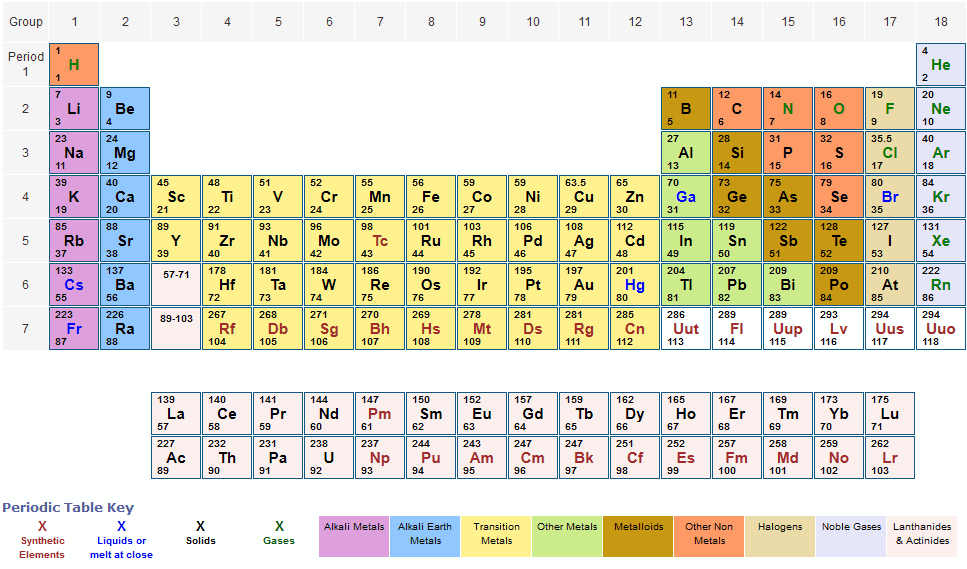
The atomic mass of elements directly relates to the concept of “mole” which is an important way of measuring the amount of substance. The average atomic mass is thus: 20.18 amu To determine the atomic mass of neon, the average of the isotopes need to be extracted: The atomic mass unit of the gas is 21.991, and the abundance of the gas is 9.25%. Neon-22: It is made up of ten protons and twelve neutrons. The atomic mass unit of the gas is 20.994, and the abundance of the gas is 0.27%. Neon-21: It is made up of ten protons and eleven neutrons. The atomic mass unit of the gas is 19.992, and the abundance of the gas is 90.48%. Neon-20: It is made up of ten protons and ten neutrons. The averaging procedure also involves taking into consideration the abundance of each isotope and multiplying it with the mass of each.Ĭonsider the gas neon, which has three isotopes: The atomic mass is determined by averaging the weight of all the isotopes of the element. The atomic mass is the average mass of the atoms of an element measured in the atomic mass unit (amu) or Daltons, D. Isotopes vary in their abundance in nature depending on the source of the materials. The most common examples are the isotopes of carbon, 12C, and 13C which possess 6 and 7 neutrons, respectively. An element (which has the same atomic number) can have multiple isotopes with different numbers of neutrons. The isotope of a given element is defined by adding the number of protons and neutrons in its nucleus. Atoms of the same element that have a different number of neutrons are called isotopes. Let us now learn the difference between atomic number and atomic mass of elements.īefore going into atomic mass, it is essential to learn about isotopes. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
This was the atomic mass of the first 30 elements. Given below is the atomic mass of elements list. Let us take a look at what is the atomic mass of elements from 1 to 30. In the same group, a periodic repetition of properties can be seen in the elements with increasing mass. Similarly, the elements in Group 8A are unreactive gaseous at room temperature. For example, the elements in Group 1A are mostly soft metals that are highly reactive with water. Elements that have similar qualities fall into the same column or group. When observing the periodic table, one can see that elements are arranged on the basis of increasing atomic numbers. The number of electrons in an element considerably determines the chemical behaviour of the element. 
The atomic number is important because the number of protons determines the number of electrons that surround the nucleus. Atoms of all elements have neutrons in the nucleus except hydrogen. Neutrons are uncharged subatomic particles which are stable when bound in an atomic nucleus. Elements are identified based on the number of protons in the nucleus regardless of the number of neutrons present. The atomic number gives a number of how many protons are inside the nucleus of the atom. The overall atomic masses that are given in periodic tables like the one for hydrogen are determined for the naturally occurring isotopes of each element, weighted by the weight of those particular isotopes on earth. Every isotope's contribution to the normal is controlled by how huge a fraction of the example it makes up. The general atomic mass is the normal of the atomic masses of the apparent multitude of various isotopes in an example. Since an element's isotopes have distinctive atomic masses, researchers may likewise decide the general atomic mass-once in a while called the atomic weight-for an element. The atomic mass number is usually rounded off to the nearest whole number. For example, a normal carbon atom with six neutrons and six protons is denoted as carbon-12. The atomic mass of a solitary atom is its absolute mass and is regularly expressed in atomic mass units or amu. Since we have seen the definition of atomic mass let us discuss it in detail. In this article, we will learn about the following things: the atomic mass of elements in detail, what is the atomic mass of all elements, and what is the atomic number and atomic mass of elements. An interesting point to note is that it is also referred to as atomic weight. The atomic mass is simply defined as the weighted average of all of the isotopes of an element, in which the mass of each isotope is multiplied by the abundance of that particular isotope. The atomic mass in Chemistry is the average mass of the atoms of an element measured in atomic mass units (amu).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
